OV329 and GABA-aminotransferase inhibition
OV329 Posters and Publications »
overview
OV329 is a next-generation GABA-aminotransferase (GABA-AT) inhibitor being developed as a potential medicine for rare and treatment-resistant forms of epilepsy and seizures. OV329 has shown to be significantly more potent than prior GABA-AT inhibitors, a strong safety profile without ophthalmic safety findings, inhibitory effects which historically demonstrate reduction in FOS frequency and potential for seizure freedom.
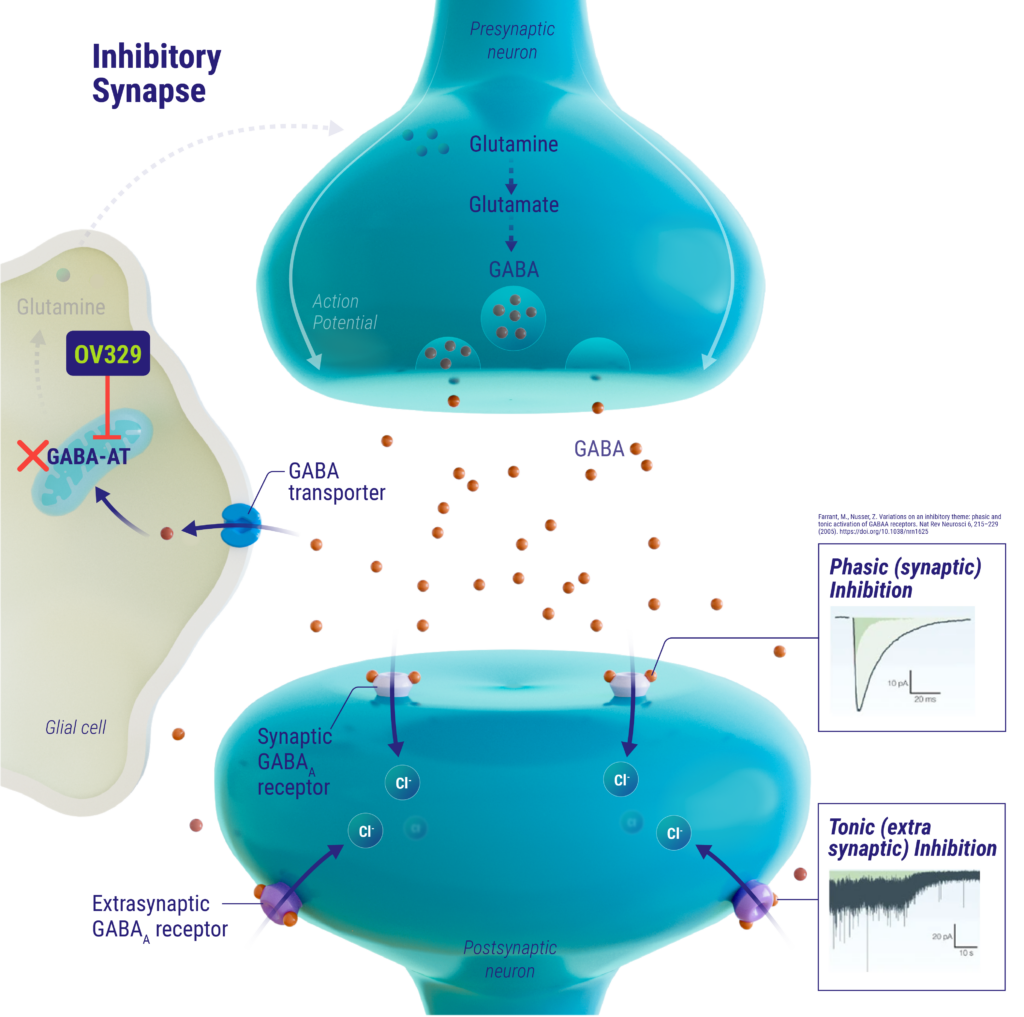
mechanism of action
γ-aminobutyric acid (GABA) is the main inhibitory neurotransmitter in the brain and is primarily catabolized by the enzyme GABA aminotransferase (GABA-AT). Low levels of GABA in the brain have been linked to neuronal hyperexcitability, which can lead to seizures. OV329 inhibits GABA-AT activity, thereby increasing GABA levels in the brain, resulting in suppression of neuronal hyperexcitability associated with seizures. OV329 may be a best-in-class GABA-AT inhibitor that could offer enhanced efficacy with an improved benefit-risk profile.
development
OV329 has completed a Phase 1 study with healthy volunteers to evaluate the safety, pharmacokinetic profile and target engagement associated with single and repeated oral doses of OV329. The Phase 1 study showed similar or better pharmacodynamic effects as vigabatrin without any ophthalmic safety findings or retinal changes associated with OV329. To date, OV329 was well-tolerated with no treatment-related SAEs. These findings support advancing into a planned Phase 2a trial.
For the OV329 topline results, webcast can be found here.
Prior evaluation of OV329 in preclinical seizure and epilepsy models indicate that OV329 significantly reduced focal and generalized seizures, including pharmaco-resistant seizure phenotypes. Improvement in seizure outcomes could be achieved with repeated, low dosing which have correlated with increases in brain GABA levels and tonic inhibitory neuronal signaling. Additionally, pre-clinical models have shown that OV329 does not preferentially accumulate in retina, eye, and brain tissues. This study demonstrated that OV329’s short half-life, quick tissue elimination properties, and prolonged pharmacodynamic effect reduced the risk of ocular accumulation occurring.
For additional information on OV329 non-clinical data, please click here.